iPSCs for Cardiac Disease Modeling |
|
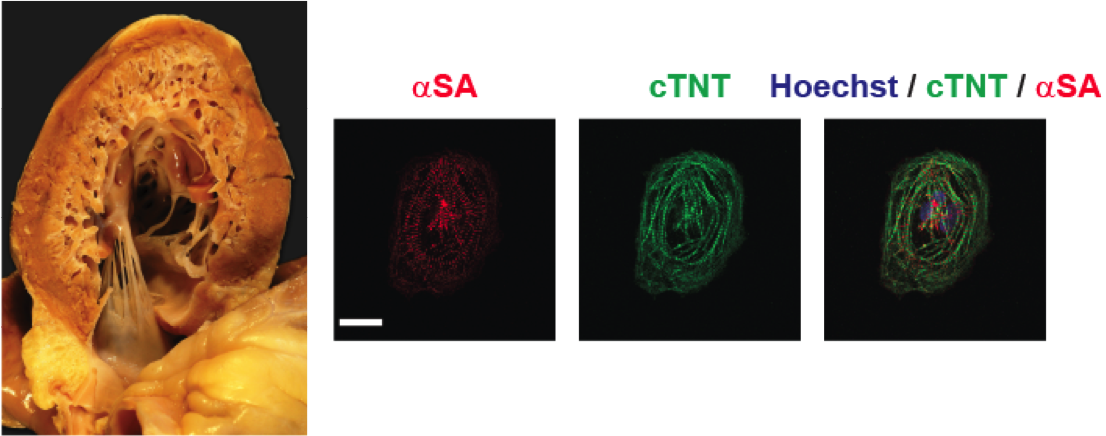
Human iPSC-derived cardiomyocytes are a unique tool for modeling cardiovascular diseases and understanding the pathobiology contributing to the development of these diseases. We recently used iPSC-derived cardiomyocytes generated from left ventricular non-compaction cardiomyopathy patients and revealed the presence of a TBX20 mutation which was associated with perturbed TGFβ signaling (Kodo & Ong, Nat Cell Biol 2016). We are currently using iPSCs to study the mechanisms leading to the development of cardiometabolic diseases with a focus on mitochondrial clearance as an adaptive mechanism. We are also actively studying the role of non-coding RNAs including snoRNAs and lncRNAs in cardiovascular diseases.
|
Regenerative Medicine & Myocardial Protection |
|
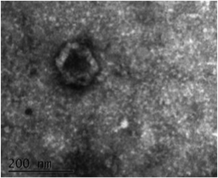
Myocardial damage in an adult heart leads to widespread loss of viable cardiomyocytes, ultimately leading to formation of scar. Current therapies are unable to offer restoration of lost cardiac tissue, leading to the development of heart failure. We seek to manipulate stem cells including cardiac progenitor cells, ESC-CMs, and iPSC-CMs as potential therapeutic strategies to address this problem (Ong et al, Circulation 2015). We also study the release of extracellular vesicles (exosomes) released by these cells which are enriched in microRNAs that may mediate myocardial repair (Ong et al, Circulation 2014, Lee et al, Stem Cells 2017).
|
We have multiple ongoing projects in the lab which address fundamental topics in cardiovascular sciences and stem cell biology and we welcome potential candidates to contact us for further discussion.